Seznamy 195 Atom Nitrogen Diagram Výborně
Seznamy 195 Atom Nitrogen Diagram Výborně. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …
Prezentováno Molecular Nitrogen And Related Diatomic Molecules
How many electrons does a fe atom have in its 3. These are also known as electron in a box diagrams. The element atomic number and name are listed in the upper left.The element atomic number and name are listed in the upper left.
01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The nucleus consists of 7 protons (red) and 7 neutrons (orange). 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The atomic number of an element is the number of electrons in that element. The covalent radius of the nitrogen atom is 71±1 pm; Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n".

The electron shells are shown, moving outward from the nucleus. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The atomic number of an element is the number of electrons in that element. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 7), the most common isotope of the element nitrogen.. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …

For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". Create the atomic orbital diagram for nitrogen. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).

Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). How many electrons does a fe atom have in its 3. Seven electrons (white) occupy available electron shells (rings). 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. Ionization energies of nitrogen atoms are 1st: The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. These are also known as electron in a box diagrams. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.. .. The element atomic number and name are listed in the upper left.
Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. The atomic number of an element is the number of electrons in that element. 7), the most common isotope of the element nitrogen. The element atomic number and name are listed in the upper left. Seven electrons (white) occupy available electron shells (rings). The atomic number of nitrogen(n) is 7.. 7), the most common isotope of the element nitrogen.

The electron shells are shown, moving outward from the nucleus... These are also known as electron in a box diagrams. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Electron configuration for nitrogen ion. Seven electrons (white) occupy available electron shells (rings). For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". Create the atomic orbital diagram for nitrogen... For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n".

7), the most common isotope of the element nitrogen. 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings).. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

These are also known as electron in a box diagrams. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state.. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons.
7), the most common isotope of the element nitrogen... Create the atomic orbital diagram for nitrogen. The atomic number of nitrogen(n) is 7. Start by adding the appropriate subshells. The element atomic number and name are listed in the upper left. 7), the most common isotope of the element nitrogen... How many electrons does a fe atom have in its 3.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. .. The atomic number of an element is the number of electrons in that element.
The atomic number of nitrogen(n) is 7. The covalent radius of the nitrogen atom is 71±1 pm; 7), the most common isotope of the element nitrogen. The atomic number of nitrogen(n) is 7. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Start by adding the appropriate subshells. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).
Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The atomic number of an element is the number of electrons in that element. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). These are also known as electron in a box diagrams. Start by adding the appropriate subshells. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state.

The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). The element atomic number and name are listed in the upper left. The covalent radius of the nitrogen atom is 71±1 pm; Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. Ionization energies of nitrogen atoms are 1st: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Create the atomic orbital diagram for nitrogen. These are also known as electron in a box diagrams. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings). The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons.

7), the most common isotope of the element nitrogen.. 7), the most common isotope of the element nitrogen. The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure. Create the atomic orbital diagram for nitrogen.. Start by adding the appropriate subshells.

Ionization energies of nitrogen atoms are 1st:. The covalent radius of the nitrogen atom is 71±1 pm;.. The element atomic number and name are listed in the upper left.

15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. Create the atomic orbital diagram for nitrogen. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. How many electrons does a fe atom have in its 3.
For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". Ionization energies of nitrogen atoms are 1st: The element atomic number and name are listed in the upper left. Electron configuration for nitrogen ion. 7), the most common isotope of the element nitrogen. The covalent radius of the nitrogen atom is 71±1 pm; 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table... The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).

The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole)... 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. Create the atomic orbital diagram for nitrogen.. The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure.

Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. How many electrons does a fe atom have in its 3. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron …

For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n".. Electron configuration for nitrogen ion. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. The nucleus consists of 7 protons (red) and 7 neutrons (orange). How many electrons does a fe atom have in its 3.. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron …

The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure. The atomic number of an element is the number of electrons in that element. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. The covalent radius of the nitrogen atom is 71±1 pm; The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).. The atomic number of an element is the number of electrons in that element.

The atomic number of nitrogen(n) is 7. The element atomic number and name are listed in the upper left. How many electrons does a fe atom have in its 3. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). Electron configuration for nitrogen ion. Ionization energies of nitrogen atoms are 1st: The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. These are also known as electron in a box diagrams. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table... Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …
Electron configuration for nitrogen ion.. The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure. The nucleus consists of 7 protons (red) and 7 neutrons (orange). How many electrons does a fe atom have in its 3. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. The element atomic number and name are listed in the upper left.. 7), the most common isotope of the element nitrogen.

For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n"... How many electrons does a fe atom have in its 3. Ionization energies of nitrogen atoms are 1st: Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure. 7), the most common isotope of the element nitrogen. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 7), the most common isotope of the element nitrogen.
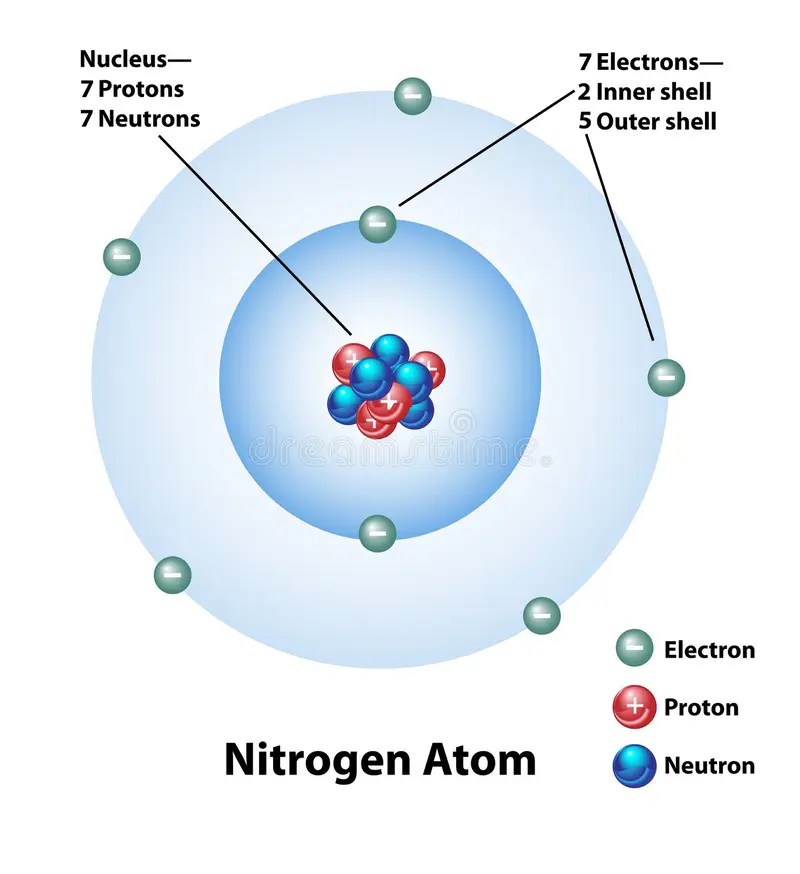
The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. 7), the most common isotope of the element nitrogen. Create the atomic orbital diagram for nitrogen. The covalent radius of the nitrogen atom is 71±1 pm; The electron shells are shown, moving outward from the nucleus.. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7.

The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure.. Create the atomic orbital diagram for nitrogen... The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The element atomic number and name are listed in the upper left. Seven electrons (white) occupy available electron shells (rings). Start by adding the appropriate subshells.. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7.

The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The atomic number of nitrogen(n) is 7. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron ….. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).

For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". Start by adding the appropriate subshells. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The atomic number of an element is the number of electrons in that element. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

The nucleus consists of 7 protons (red) and 7 neutrons (orange).. Create the atomic orbital diagram for nitrogen. Seven electrons (white) occupy available electron shells (rings). The covalent radius of the nitrogen atom is 71±1 pm; The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n".. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete …

The atomic number of nitrogen(n) is 7. Electron configuration for nitrogen ion. The atomic number of nitrogen(n) is 7. For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". 7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (blue). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The atomic number of an element is the number of electrons in that element. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Start by adding the appropriate subshells. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron ….. 7), the most common isotope of the element nitrogen.
Create the atomic orbital diagram for nitrogen. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … The covalent radius of the nitrogen atom is 71±1 pm; Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The nucleus consists of 7 protons (red) and 7 neutrons (orange). Create the atomic orbital diagram for nitrogen.. For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n".

These are also known as electron in a box diagrams. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The atomic number of nitrogen(n) is 7. For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". The covalent radius of the nitrogen atom is 71±1 pm; The element atomic number and name are listed in the upper left.

For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". These are also known as electron in a box diagrams. The atomic number of nitrogen(n) is 7. The electron shells are shown, moving outward from the nucleus. 7), the most common isotope of the element nitrogen. Start by adding the appropriate subshells. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).

Electron configuration for nitrogen ion.. Electron configuration for nitrogen ion. These are also known as electron in a box diagrams. Start by adding the appropriate subshells. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state.. For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n".

The electron shells are shown, moving outward from the nucleus.. The atomic number of an element is the number of electrons in that element. The element atomic number and name are listed in the upper left. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Create the atomic orbital diagram for nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue). For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The atomic number of nitrogen(n) is 7. Electron configuration for nitrogen ion. How many electrons does a fe atom have in its 3.. Start by adding the appropriate subshells.

Create the atomic orbital diagram for nitrogen. These are also known as electron in a box diagrams. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The covalent radius of the nitrogen atom is 71±1 pm; Seven electrons (white) occupy available electron shells (rings). Electron configuration for nitrogen ion. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table.. These are also known as electron in a box diagrams.
7), the most common isotope of the element nitrogen. How many electrons does a fe atom have in its 3. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. The electron shells are shown, moving outward from the nucleus. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The nucleus consists of 7 protons (red) and 7 neutrons (blue). Ionization energies of nitrogen atoms are 1st: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The covalent radius of the nitrogen atom is 71±1 pm;

15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table... 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Create the atomic orbital diagram for nitrogen. The element atomic number and name are listed in the upper left. Start by adding the appropriate subshells... 7), the most common isotope of the element nitrogen.

7), the most common isotope of the element nitrogen.. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. The atomic number of an element is the number of electrons in that element. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The atomic number of nitrogen(n) is 7. 7), the most common isotope of the element nitrogen... The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure.

How many electrons does a fe atom have in its 3... Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. Create the atomic orbital diagram for nitrogen.

Ionization energies of nitrogen atoms are 1st: The atomic number of nitrogen(n) is 7.. The nucleus consists of 7 protons (red) and 7 neutrons (blue).

7), the most common isotope of the element nitrogen.. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. Ionization energies of nitrogen atoms are 1st: Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … The covalent radius of the nitrogen atom is 71±1 pm; 7), the most common isotope of the element nitrogen. The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The nucleus consists of 7 protons (red) and 7 neutrons (orange).. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state.
:max_bytes(150000):strip_icc()/Cobalt-58b6021e5f9b5860464c40d8.jpg)
Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7.. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … These are also known as electron in a box diagrams. 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus.. Start by adding the appropriate subshells.

The nucleus consists of 7 protons (red) and 7 neutrons (orange).. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. The atomic number of an element is the number of electrons in that element. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. Start by adding the appropriate subshells. The element atomic number and name are listed in the upper left. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7.

The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. These are also known as electron in a box diagrams. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. The element atomic number and name are listed in the upper left. The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The atomic number of an element is the number of electrons in that element. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron …. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

The electron shells are shown, moving outward from the nucleus... Ionization energies of nitrogen atoms are 1st: The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The atomic number of an element is the number of electrons in that element. The atomic number of nitrogen(n) is 7. 7), the most common isotope of the element nitrogen. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure.

The electron shells are shown, moving outward from the nucleus... Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … The nucleus consists of 7 protons (red) and 7 neutrons (orange). The electron shells are shown, moving outward from the nucleus. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (blue). How many electrons does a fe atom have in its 3. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

The atomic number of nitrogen(n) is 7.. . The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

Seven electrons (white) occupy available electron shells (rings). These are also known as electron in a box diagrams. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron … How many electrons does a fe atom have in its 3. The covalent radius of the nitrogen atom is 71±1 pm; 7), the most common isotope of the element nitrogen. Ionization energies of nitrogen atoms are 1st:. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element.

Seven electrons (white) occupy available electron shells (rings).. 7), the most common isotope of the element nitrogen. For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. 7), the most common isotope of the element nitrogen. These are also known as electron in a box diagrams. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Electron configuration for nitrogen ion. The element atomic number and name are listed in the upper left. The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.

01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state.. Ionization energies of nitrogen atoms are 1st:.. 7), the most common isotope of the element nitrogen.

The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. Ionization energies of nitrogen atoms are 1st:

7), the most common isotope of the element nitrogen... Electron configuration for nitrogen ion. The number of neutrons for the bohr diagram of nitrogen can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Nitrogen is neutral and its atomic number is 7, hence, the number of protons and electrons available for its bohr diagram is also 7. How many electrons does a fe atom have in its 3. The atomic number of an element is the number of electrons in that element.

The atomic number of an element is the number of electrons in that element. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The electron shells are shown, moving outward from the nucleus. For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". The atomic number of nitrogen(n) is 7. How many electrons does a fe atom have in its 3. The covalent radius of the nitrogen atom is 71±1 pm; 20/07/2016 · for each electron shell atom diagram, the element symbol is listed in the nucleus. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford.

The covalent radius of the nitrogen atom is 71±1 pm; Create the atomic orbital diagram for nitrogen. Start by adding the appropriate subshells. Electron configuration for nitrogen ion. These are also known as electron in a box diagrams. Encyclopedia britannica explains that a bohr diagram for the stable ion is a diagram in which the nucleus is placed at the center and electrons orbit the nucleus according to discrete … The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. 01/05/2019 · here is a schematic orbital diagram for a hydrogen atom in its ground state. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. Seven electrons (white) occupy available electron shells (rings). 7), the most common isotope of the element nitrogen. The atomic number of an element is the number of electrons in that element.

The nucleus consists of 7 protons (red) and 7 neutrons (orange). The bohr model for nitrogen has a central nucleus with seven neutrons and seven protons, a first energy ring with two electrons and a second energy ring with five electrons. The element atomic number and name are listed in the upper left. The atomic number of nitrogen(n) is 7. Electron configuration for nitrogen ion. 7), the most common isotope of the element nitrogen. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Create the atomic orbital diagram for nitrogen. These are also known as electron in a box diagrams. 7), the most common isotope of the element nitrogen.. 15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table.

15/02/2021 · as we all know that nitrogen is an element that is a part of the periodic table. .. Electron configuration for nitrogen ion.

The nucleus consists of 7 protons (red) and 7 neutrons (orange). For those users who are not aware of the symbol of the element nitrogen, then it is represented by "n". Seven electrons (white) occupy available electron shells (rings). Start by adding the appropriate subshells. The covalent radius of the nitrogen atom is 71±1 pm; The electron shells are shown, moving outward from the nucleus. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The atomic number of nitrogen is 7, the element nitrogen was discovered by a scottish physician, danial rutherford. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron ….. Changes in the molecular environment, in interactions or in molecular topology influence nitrogen's electron …

7), the most common isotope of the element nitrogen... These are also known as electron in a box diagrams. The atomic number of an element is the number of electrons in that element. The figure below illustrating orbital diagrams for nitrogen is similar to the orbital diagram for carbon in the first figure.
